HALAVEN® demonstrated a significant survival benefit in a Phase III study of patients with advanced liposarcoma1
Treatment effects of HALAVEN were demonstrated in patients with advanced liposarcoma based on preplanned, exploratory subgroup analyses of overall survival and progression-free survival2
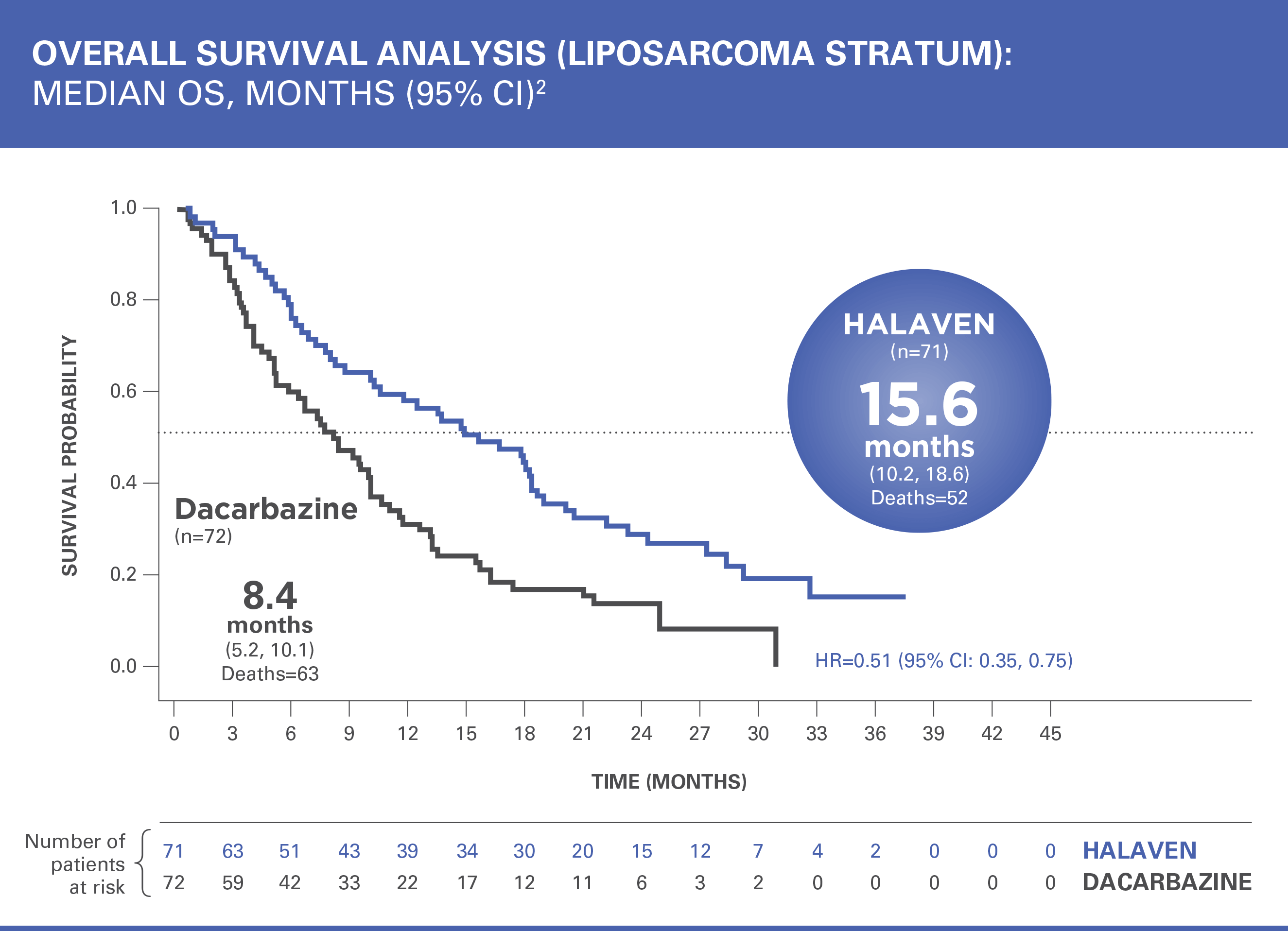
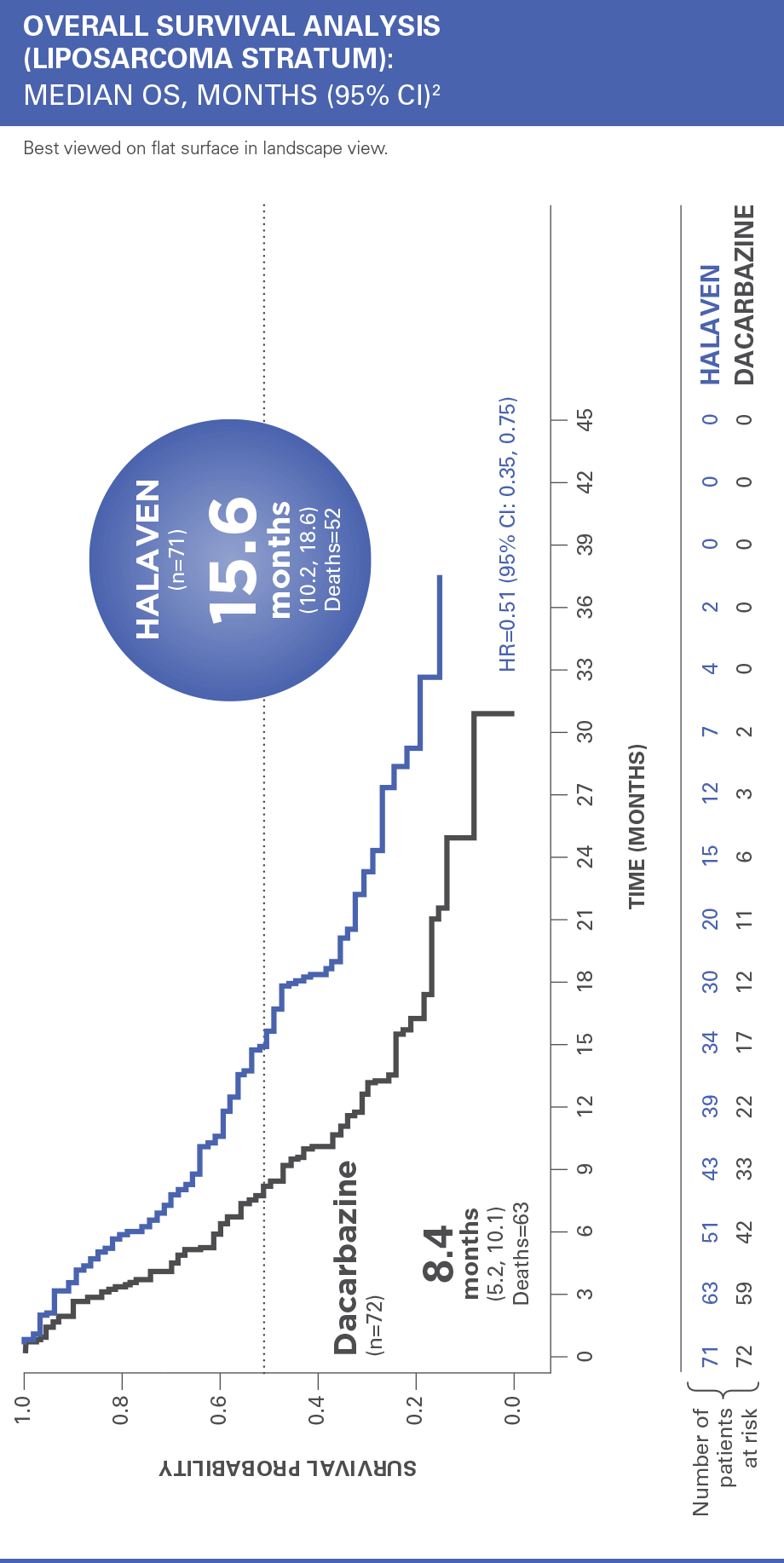
The efficacy and safety of HALAVEN were evaluated in an open-label, randomized (1:1), multicenter, active-controlled trial. Eligible patients were required to have unresectable, locally advanced, or metastatic liposarcoma or leiomyosarcoma, at least 2 prior systemic chemotherapies (one of which must have included an anthracycline), and disease progression within 6 months of the most recent chemotherapy regimen. Patients were randomized to HALAVEN 1.4 mg/m2 administered intravenously on Days 1 and 8 of a 21-day cycle or to dacarbazine at a dose of 850 mg/m2, 1,000 mg/m2, or 1,200 mg/m2 administered intravenously every 21 days (dacarbazine dose was selected by the investigator prior to randomization). Treatment continued until disease progression or unacceptable toxicity. Randomization was stratified by histology (liposarcoma or leiomyosarcoma), number of prior therapies (2 vs >2), and geographic region. The most common (>40%) prior systemic chemotherapies were doxorubicin (90%), ifosfamide (62%), gemcitabine (59%), trabectedin (50%), and docetaxel (48%).2
OS=overall survival; CI=confidence interval.
-
There was no evidence of efficacy of HALAVEN in patients with advanced or metastatic leiomyosarcoma in this trial2
Patients in the HALAVEN arm with liposarcoma had a 49% reduction in relative risk of death vs control group2,3
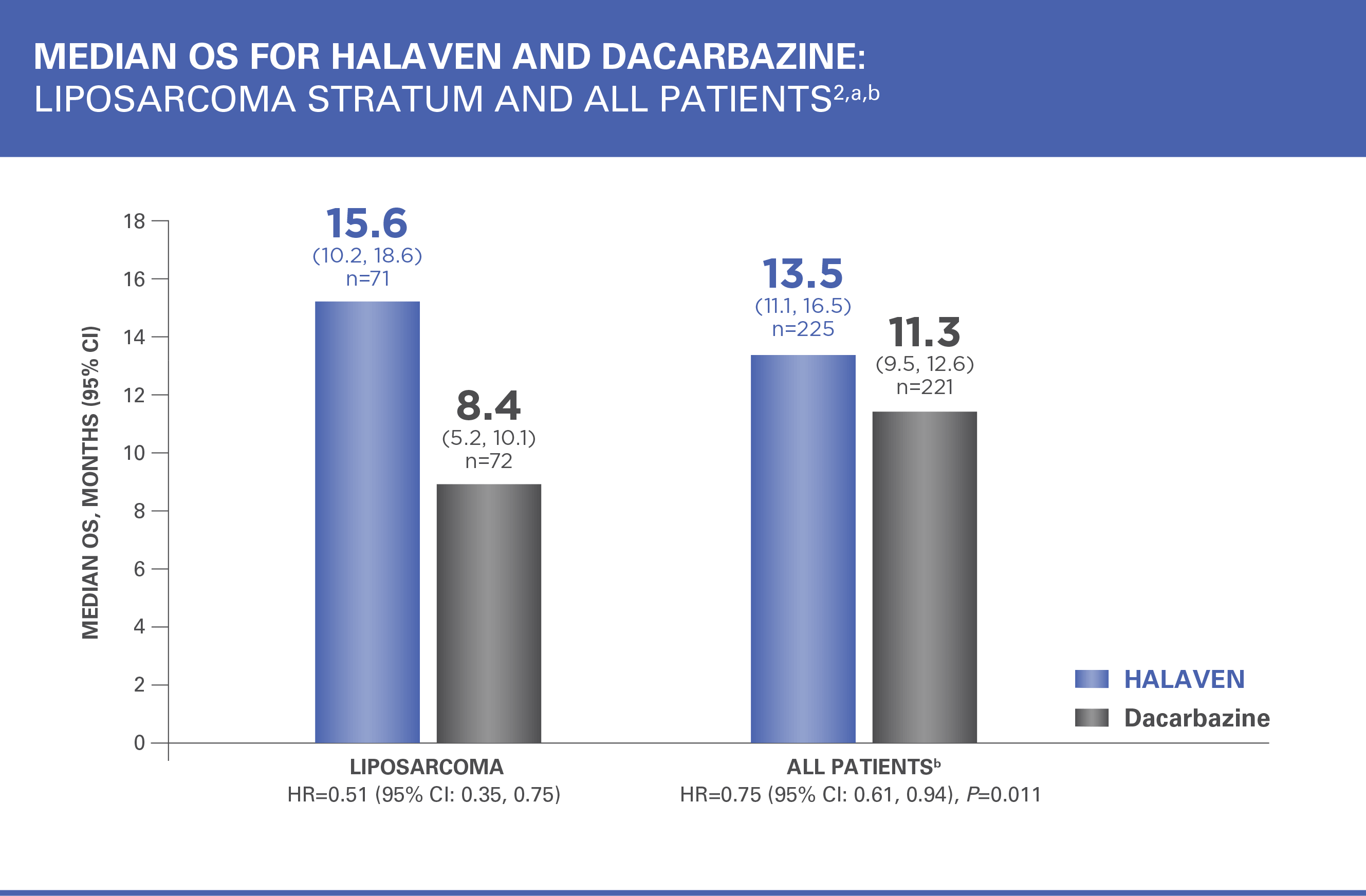
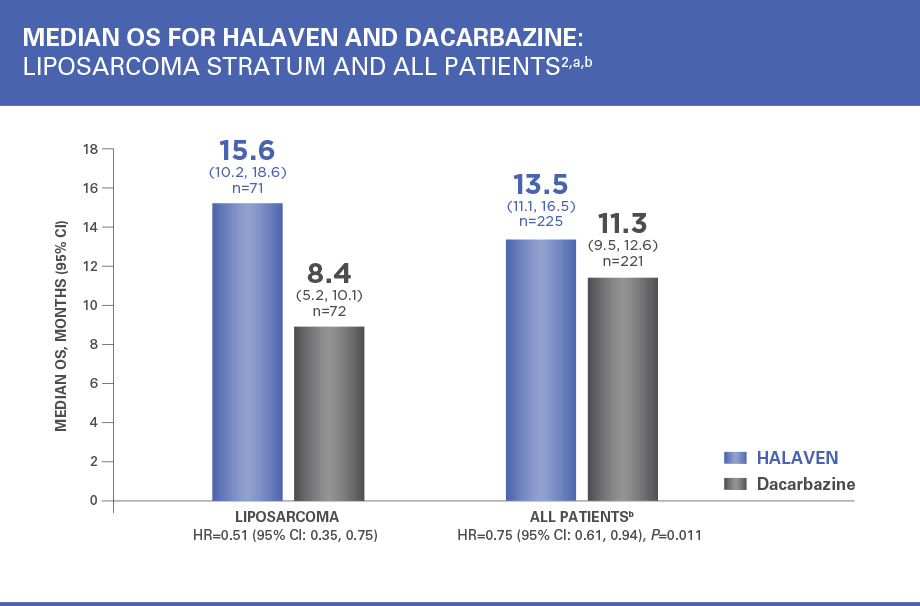
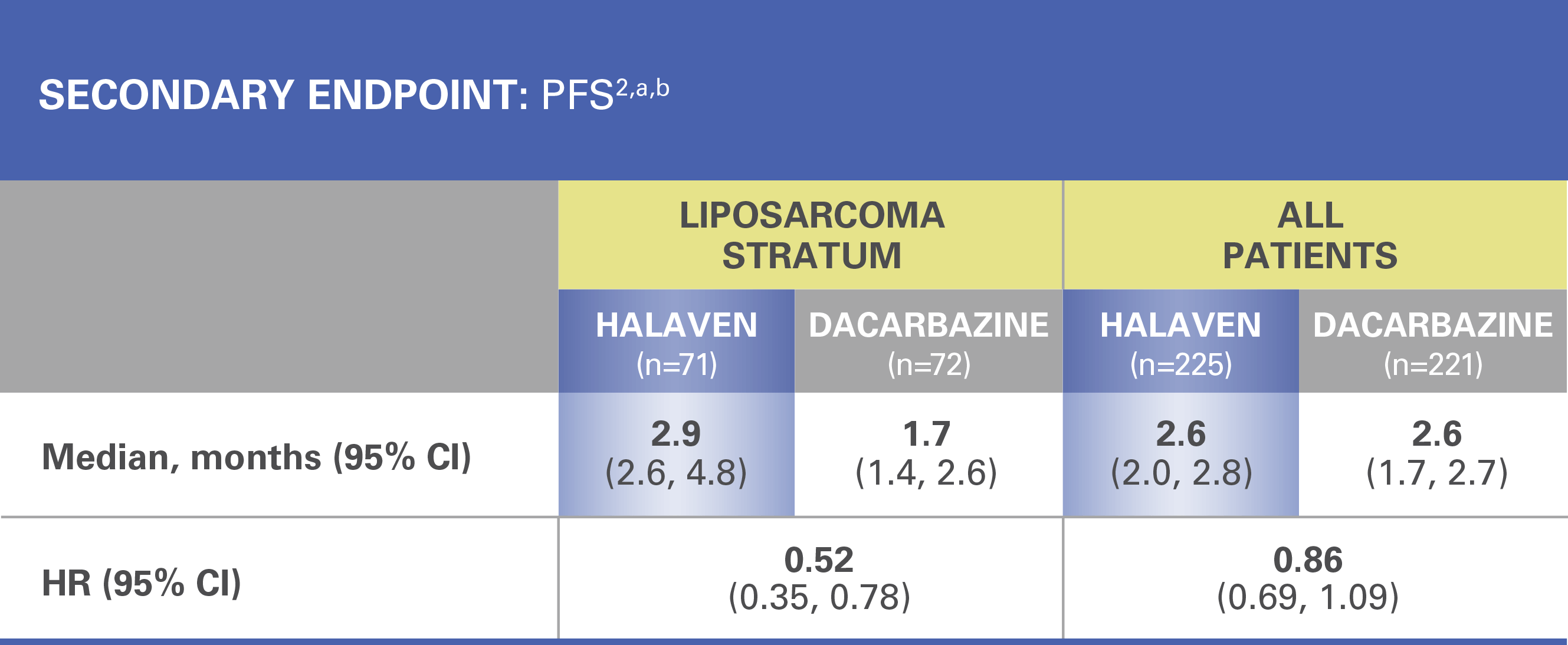
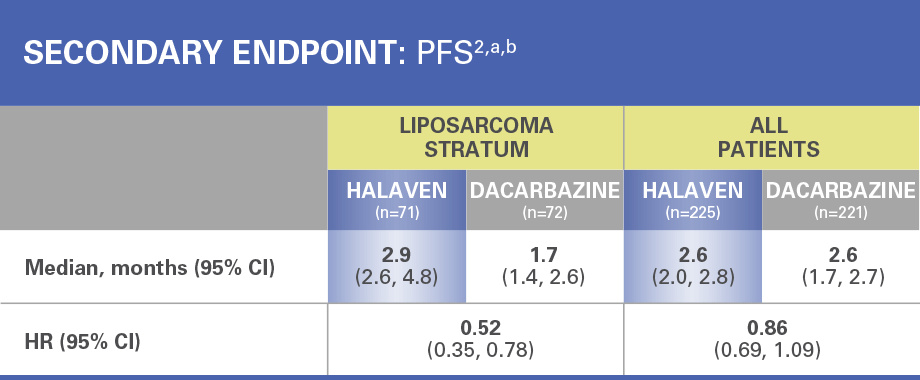
HR=hazard ratio; PFS=progression-free survival.
aEfficacy data from 1 study site enrolling 6 patients were excluded.
bAll patients=liposarcoma and leiomyosarcoma.
Indications
Metastatic Breast Cancer
HALAVEN (eribulin mesylate) Injection is indicated for the treatment of patients with metastatic breast cancer (mBC) who have previously received at least 2 chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting.
Liposarcoma
HALAVEN is indicated for the treatment of patients with unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen.
Important Safety Information
Warnings and Precautions
Neutropenia: Severe neutropenia (ANC <500/mm3) lasting >1 week occurred in 12% of patients with mBC and liposarcoma or leiomyosarcoma. Febrile neutropenia occurred in 5% of patients with mBC and 2 patients (0.4%) died from complications. Febrile neutropenia occurred in 0.9% of patients with liposarcoma or leiomyosarcoma, and fatal neutropenic sepsis occurred in 0.9% of patients. Patients with mBC with elevated liver enzymes >3 × ULN and bilirubin >1.5 × ULN experienced a higher incidence of Grade 4 neutropenia and febrile neutropenia than patients with normal levels. Monitor complete blood cell counts prior to each dose, and increase the frequency of monitoring in patients who develop Grade 3 or 4 cytopenias. Delay administration and reduce subsequent doses in patients who experience febrile neutropenia or Grade 4 neutropenia lasting >7 days.
Peripheral Neuropathy: Grade 3 peripheral neuropathy occurred in 8% of patients with mBC (Grade 4=0.4%) and 22% developed a new or worsening neuropathy that had not recovered within a median follow-up duration of 269 days (range 25-662 days). Neuropathy lasting >1 year occurred in 5% of patients with mBC. Grade 3 peripheral neuropathy occurred in 3.1% of patients with liposarcoma and leiomyosarcoma receiving HALAVEN and neuropathy lasting more than 60 days occurred in 58% (38/65) of patients who had neuropathy at the last treatment visit. Patients should be monitored for signs of peripheral motor and sensory neuropathy. Withhold HALAVEN in patients who experience Grade 3 or 4 peripheral neuropathy until resolution to Grade 2 or less.
Embryo-Fetal Toxicity: HALAVEN can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during treatment with HALAVEN and for at least 2 weeks following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with HALAVEN and for 3.5 months following the final dose.
QT Prolongation: Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Correct hypokalemia or hypomagnesemia prior to initiating HALAVEN and monitor these electrolytes periodically during therapy. Avoid in patients with congenital long QT syndrome.
Adverse Reactions
In patients with mBC receiving HALAVEN, the most common adverse reactions (≥25%) were neutropenia (82%), anemia (58%), asthenia/fatigue (54%), alopecia (45%), peripheral neuropathy (35%), nausea (35%), and constipation (25%). Febrile neutropenia (4%) and neutropenia (2%) were the most common serious adverse reactions. The most common adverse reaction resulting in discontinuation was peripheral neuropathy (5%).
In patients with liposarcoma and leiomyosarcoma receiving HALAVEN, the most common adverse reactions (≥25%) reported in patients receiving HALAVEN were fatigue (62%), nausea (41%), alopecia (35%), constipation (32%), peripheral neuropathy (29%), abdominal pain (29%), and pyrexia (28%). The most common (≥5%) Grade 3-4 laboratory abnormalities reported in patients receiving HALAVEN were neutropenia (32%), hypokalemia (5.4%), and hypocalcemia (5%). Neutropenia (4.9%) and pyrexia (4.5%) were the most common serious adverse reactions. The most common adverse reactions resulting in discontinuation were fatigue and thrombocytopenia (0.9% each).
Use in Specific Populations
Lactation: Because of the potential for serious adverse reactions in breastfed infants from eribulin mesylate, advise women not to breastfeed during treatment with HALAVEN and for 2 weeks after the final dose.
Hepatic and Renal Impairment: A reduction in starting dose is recommended for patients with mild or moderate hepatic impairment and/or moderate or severe renal impairment.
Indications
Metastatic Breast Cancer
HALAVEN (eribulin mesylate) Injection is indicated for the treatment of patients with metastatic breast cancer (mBC) who have previously received at least 2 chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting.
Liposarcoma
HALAVEN is indicated for the treatment of patients with unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen.
Indications
Metastatic Breast Cancer
HALAVEN (eribulin mesylate) Injection is indicated for the treatment of patients with metastatic breast cancer (mBC) who have previously received at least 2 chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting.
Liposarcoma
HALAVEN is indicated for the treatment of patients with unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen.
Important Safety Information
Warnings and Precautions
Neutropenia: Severe neutropenia (ANC <500/mm3) lasting >1 week occurred in 12% of patients with mBC and liposarcoma or leiomyosarcoma. Febrile neutropenia occurred in 5% of patients with mBC and 2 patients (0.4%) died from complications. Febrile neutropenia occurred in 0.9% of patients with liposarcoma or leiomyosarcoma, and fatal neutropenic sepsis occurred in 0.9% of patients. Patients with mBC with elevated liver enzymes >3 × ULN and bilirubin >1.5 × ULN experienced a higher incidence of Grade 4 neutropenia and febrile neutropenia than patients with normal levels. Monitor complete blood cell counts prior to each dose, and increase the frequency of monitoring in patients who develop Grade 3 or 4 cytopenias. Delay administration and reduce subsequent doses in patients who experience febrile neutropenia or Grade 4 neutropenia lasting >7 days.
Peripheral Neuropathy: Grade 3 peripheral neuropathy occurred in 8% of patients with mBC (Grade 4=0.4%) and 22% developed a new or worsening neuropathy that had not recovered within a median follow-up duration of 269 days (range 25-662 days). Neuropathy lasting >1 year occurred in 5% of patients with mBC. Grade 3 peripheral neuropathy occurred in 3.1% of patients with liposarcoma and leiomyosarcoma receiving HALAVEN and neuropathy lasting more than 60 days occurred in 58% (38/65) of patients who had neuropathy at the last treatment visit. Patients should be monitored for signs of peripheral motor and sensory neuropathy. Withhold HALAVEN in patients who experience Grade 3 or 4 peripheral neuropathy until resolution to Grade 2 or less.
Embryo-Fetal Toxicity: HALAVEN can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during treatment with HALAVEN and for at least 2 weeks following the final dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with HALAVEN and for 3.5 months following the final dose.
QT Prolongation: Monitor for prolonged QT intervals in patients with congestive heart failure, bradyarrhythmias, drugs known to prolong the QT interval, and electrolyte abnormalities. Correct hypokalemia or hypomagnesemia prior to initiating HALAVEN and monitor these electrolytes periodically during therapy. Avoid in patients with congenital long QT syndrome.
Adverse Reactions
In patients with mBC receiving HALAVEN, the most common adverse reactions (≥25%) were neutropenia (82%), anemia (58%), asthenia/fatigue (54%), alopecia (45%), peripheral neuropathy (35%), nausea (35%), and constipation (25%). Febrile neutropenia (4%) and neutropenia (2%) were the most common serious adverse reactions. The most common adverse reaction resulting in discontinuation was peripheral neuropathy (5%).
In patients with liposarcoma and leiomyosarcoma receiving HALAVEN, the most common adverse reactions (≥25%) reported in patients receiving HALAVEN were fatigue (62%), nausea (41%), alopecia (35%), constipation (32%), peripheral neuropathy (29%), abdominal pain (29%), and pyrexia (28%). The most common (≥5%) Grade 3-4 laboratory abnormalities reported in patients receiving HALAVEN were neutropenia (32%), hypokalemia (5.4%), and hypocalcemia (5%). Neutropenia (4.9%) and pyrexia (4.5%) were the most common serious adverse reactions. The most common adverse reactions resulting in discontinuation were fatigue and thrombocytopenia (0.9% each).
Use in Specific Populations
Lactation: Because of the potential for serious adverse reactions in breastfed infants from eribulin mesylate, advise women not to breastfeed during treatment with HALAVEN and for 2 weeks after the final dose.
Hepatic and Renal Impairment: A reduction in starting dose is recommended for patients with mild or moderate hepatic impairment and/or moderate or severe renal impairment.
References: 1. Schöffski P, Chawla S, Maki RG, et al. Eribulin versus dacarbazine in previously treated patients with advanced liposarcoma or leiomyosarcoma: a randomised, open-label, multicentre, phase 3 trial. Lancet. 2016;387(10028):1629-1637. 2. HALAVEN [package insert]. Nutley, NJ: Eisai Inc. 3. NCI dictionary of cancer terms. National Cancer Institute Web site. http://www.cancer.gov/publications/dictionaries/cancer-terms?CdrID=618612. Accessed January 15, 2016.

